|
In terms of cellular potency, ontogenesis is considered a one-way street with a continuous loss of potency from the totipotent zygote (and very early blastomere stages) to pluripotent ICM and then to less potent cells (multi-, di- and unipotent cells). With further improvements, such as the reduction of heterogeneity in TPS cells as well as optimizing the induction protocol to guide the precise self-differentiation and organization capability of TPS cells during the formation of blastoids, TPS cells might be able to reconstitute a fully functional synthetic embryo in the future.The first successful isolation and maintenance of ES derived from the inner cell mass (ICM) of murine blastocyst stages was described in 1981, 2 and since then acted as game changer for genetic studies in this mammalian model organism. Although the current TPS cells are not capable of generating fully functional blastoids, TPS-blastoids seem to outperform previous attempts and generate cell lineages very close to those in the blastocyst. presents a valuable resource that can potentially be applied in human or other mammalian species for totipotency capture and has a broad application in regenerative medicine. 6, 7 We envision that the study by Xu et al. Recently, two studies reported the existence of a small subpopulation of 8-cell-like cells in cultured naïve human PSCs, which can be enhanced by epigenetic modulation. Taken together, these observations suggest that TPS cells have the potential to differentiate into both embryonic and extraembryonic lineages both in vitro and in vivo.īeyond the mouse, in vitro capture of human 2- or 8-cell-like cell type has attracted a lot of interest for years. TPS-blastoids were able to implant and induce decidualization after transferring into a surrogate. It is worthy of mentioning, compared to EPS-blastoids, 5 EPS-converted TPS-blastoids contain much fewer intermediate cells, which further confirms the superior potency of TPS cells on lineage differentiation. Moreover, by comparing with blastoids generated by other methods, the author found that trophectoderm cells in TPS-blastoids resemble preimplantation trophectoderm in blastocyst instead of postimplantation extraembryonic ectoderm. Single-cell RNA-seq data showed that the TPS-blastoid cells cluster well with the respective cell lineages from blastocysts. To further investigate the potential of TPS cells, the authors attempted the formation of blastocyst-like structures (blastoids) from TPS cells and successfully generated blastoids with both epiblast and trophectoderm markers expression. The author substantiated this finding by ruling out the possibility of having artificially fused cells of TPS cells and host cells in the placenta using differentially labeled host and injected TPS cells. Marker expression and single-cell RNA-seq analysis at early (E10.5) and late (E17.5) stages revealed a high contribution of TPS cells in the placenta. The resulting chimeric embryos showed germline contribution of TPS cells and the presence of TPS-derived trophoblast and visceral endoderm cells. 
For this, the authors designed and performed chimera formation assays with multiple cells or more stringently, just as a single cell injected into 8-cell embryos. 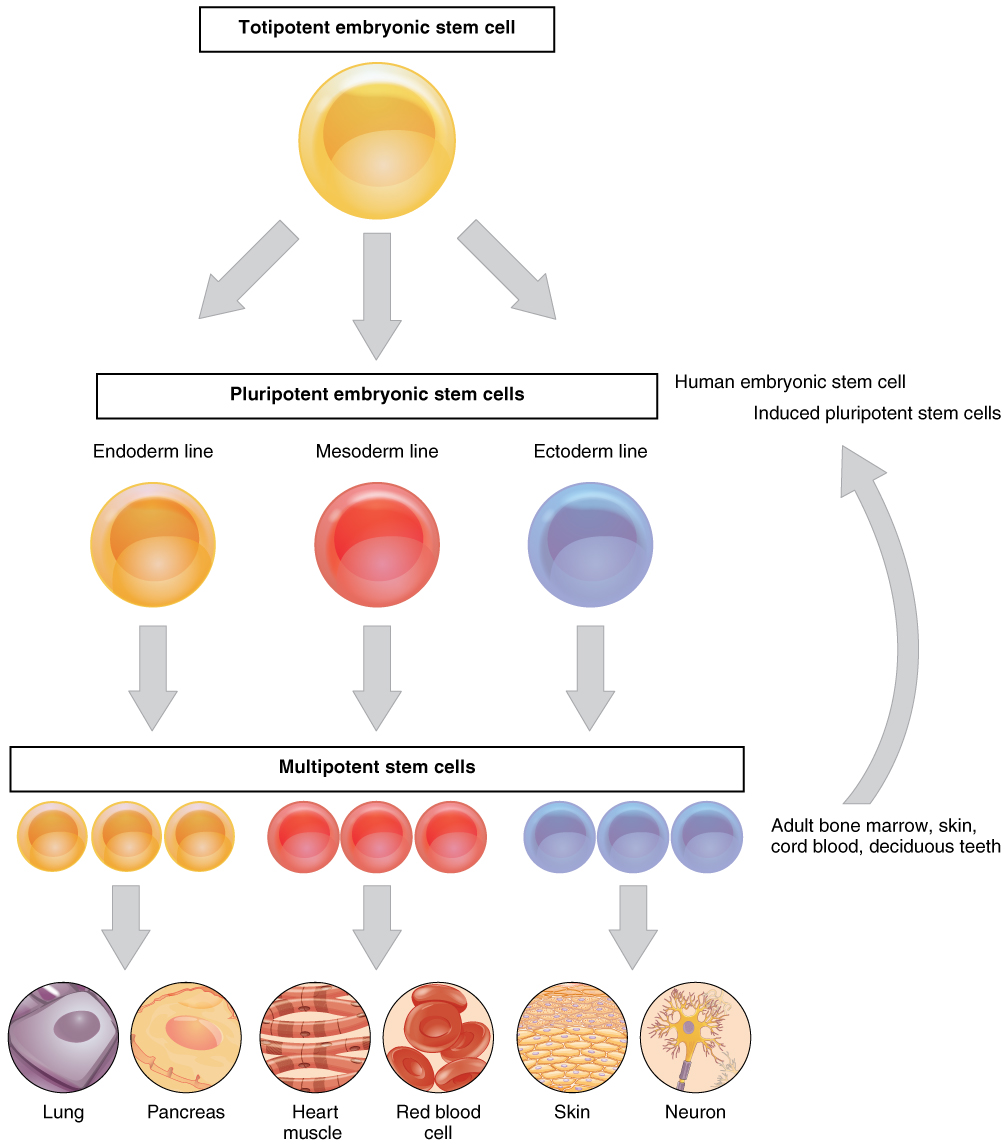
To further substantiate their claims in vivo, the authors examined whether TPS cells have an unbiased ability to contribute to both embryonic and extraembryonic lineage development. Importantly, the cocktail can be successfully employed to derive totipotent-like cells directly from 2-cell mouse embryos without any adaption step. Besides several molecular totipotent features, CPEC-converted cells are also capable of differentiating into extraembryonic cells in vitro. 3 The finding that the CPEC condition as a whole supports totipotency is interesting and novel. Interestingly, RA signaling was recently identified as a critical regulator of totipotency, 4 and DOT1L inhibition was also used in another study as a component for sustaining totipotency in vitro. A condition containing CD1530 (a RARγ agonist), V PA (an HDAC inhibitor), EPZ004777 (a DOT1L inhibitor) and CHIR99021 (a WNT agonist) (collectively, the CPEC condition) turns out to largely and stably induce and maintain cells preserving the totipotent molecular features with intense expression of totipotency markers. 3 performed a chemical screen in extended pluripotent stem (EPS) cells and identified chemicals that can significantly increase 2-cell marker (MERVL-tdTomato/Zscan4-GFP) expression and positive subpopulation. 
In June issue of Cell Research, Xu et al.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
